SBU Conducting Study to Find Cure for COVID-19 Patients
Centerport Resident Among First to Donate Convalescent Blood Plasma

Stony Brook Medicine has launched a research study in the hopes of developing a treatment for those severely suffering from the coronavirus.
On April 2, SBM began a U.S. Food and Drug Administration-approved research study to determine if convalescent blood plasma from those who’ve recovered from COVID-19 can help treat currently hospitalized patients. One of the first volunteers was Mark Goidell, a litigation attorney from Centerport.
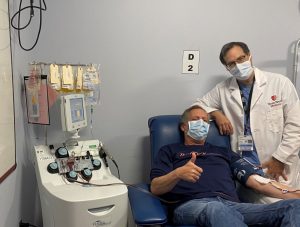
The Research Study
Dr. Elliott Bennett-Guerrero, vice chair of Clinical Research and Innovation in the Renaissance School of Medicine Department of Anesthesiology, is heading up the research study. He said the hospital needs approximately 100 volunteers who have recovered from the coronavirus to donate blood plasma, possibly once a week or every other week. Subjects must have contracted COVID-19 and be free of symptoms for 14 days. As of April 22, the doctor said they have received a large number of inquiries leading to 180 people being screened and 90 have been identified as having high levels of antibodies. Currently 25 have either donated blood plasma or are scheduled to do so.
Bennett-Guerrero said researchers are looking for those with high antibody levels of the virus and testing takes about 15 minutes. The donors must also meet regular criteria to be a blood donor, the doctor said, which includes being at least 17 years old, weighing more than 100 pounds, and having no infections Certain travel outside of the U.S. will also be reviewed.
“We’re very fortunate that we can run this protocol independently, because we have access to a very good test for antibodies, and we also have a licensed blood collection facility already in our hospital,” the doctor said. “So we have those two main ingredients to help us to collect blood plasma and unfortunately have a large number of patients who are in desperate need of help.”
Bennett-Guerrero said the trial will include 500 hospital patients ranging from those who are intubated and those who are not. A higher percentage of patients will receive convalescent serum on a random basis compared to other trials which tend to have 50 percent of patients serve as a control group who receive a placebo.
“Our protocol is unique in that while we want to help as many people as possible, we also want to determine if it’s safe and effective,” the doctor said. “It’s a randomized trial where 80 percent of the patients will receive the convalescent plasma because we hope to benefit as many patients as possible, and there will be a small group of 20 percent of patients that will serve as the control group and get standard plasma. It’s the only way we can rigorously determine if it’s safe and effective to do this.”
Plasma, which is the liquid portion of the blood, helps with clotting and supporting immunity. The hope is the plasma from those who have survived COVID-19 will contain antibodies which in turn can kill the virus in seriously ill patients. According to SBM, convalescent serum therapy is a century-old treatment that has been used in patients during the Spanish Flu pandemic of 1918, the diphtheria epidemic in the U.S. in the 1920s, and more recently, the Ebola outbreak in 2014.
The doctor said it’s too early to determine if giving convalescent blood plasma to a COVID-19 patient will help.
“It’s very early in the stage with this pandemic,” he said. “We’re only beginning to learn what are the patterns of antibody formation in people who had the COVID-19 infection. In general it’s believed that antibodies to COVID-19 will probably persist for a while, perhaps months or years, and likely be protective. However, we don’t know yet if the antibodies that we are measuring actually mean, ‘quote-unquote,’ one is immune and can’t be reinfected. We think that’s probably the case but it’s not proven yet.”

The Donor
The doctor said Goidell was a good candidate because he was free of symptoms for a couple of weeks, had high levels of the antibodies in his system and met blood donation criteria.
Both Goidell, 64, and his wife Lynn, 62, came down with the virus. The attorney said he was sick toward the end of February and in early March, and his symptoms included being lethargic and feverish, and at times during the night he would frequently wake up and try to catch his breath, many times going outside to do so.
His wife was admitted to Huntington Hospital March 13 due to having double pneumonia and was discharged a few days later. Goidell said he did have a relapse where he said his symptoms felt like a sinus infection, with a loss of smell and taste. He said he has recovered about 70 percent of those senses.
While his symptoms didn’t initially lead to testing, he said, once his wife was hospitalized he was tested March 17 at an urgent care facility. After reading about the Stony Brook study on the News12 website, Goidell said he was more than willing to participate in the trial.
“It’s heartbreaking to see what’s happening and all the tragedy and anguish that is being brought about by the virus,” Goidell said. “I’m grateful for the fact that I’ve recovered, and I’m able to do something to help.”
He said he feels fortunate to live in close proximity to Stony Brook Medicine. Between his experience with the study so far and his wife’s hospital stay at Huntington Hospital, he has gained an even greater respect and admiration for health care workers. He called those who treated his wife “heroes.”
He added the two of them are now back to working remotely, joking that he has put on some weight due to his wife’s good cooking, and he has been playing a lot of basketball in his driveway to burn off the pounds.
He said he hopes that others who have recovered will donate their plasma, and that others will “stay inside and help each other out.”
“I wish Dr. Bennett-Guerrero and the researchers at Stony Brook the best of luck, and I have the most gratitude for the work they are doing,” he said.
People who have recovered from COVID-19 and want to donate blood plasma can visit www.stonybrookmedicine.edu/COVID_donateplasma where they will be required to fill out an online survey. Potentially eligible people will be asked to participate in a screening visit at a Stony Brook Medicine facility, which will take approximately 30 minutes. You do not need to be a Stony Brook University Hospital patient to participate, but you must meet the required criteria for plasma donation and have high levels of antibodies to the virus that causes COVID-19.






